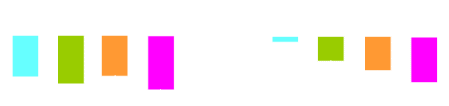
RIO programme: placebo-subtracted change for multiple CMR
factors
SBP (mmHg)
Waist circumference (cm)
Triglycerides (%)
HDL-cholesterol (%)
*
*
*
*
%
*p<0.001




*
*
*
*
*p<0.001




*
*p<0.001
cm




-3.6
*
-4.2
*
-4.7
*
-3.3
0
-1
-2
-3
-4
-5
-6
p<0.05
p<0.05
NS
NS





-0.2
-1.2
-1.7
-2.3
mmHg
0
-0.5
-1
-1.5
-2
-2.5
-3
0.5
↓ HbA1C
Pi-Sunyer FX et al, 2006;
Després JP et al, 2005; Van Gaal L et al, 2005









-20
-15
-10
-5
0
5
10
7.2
8.9
8.1
8.4










-6
-5
-4
-3
-2
-1
0
-
13.2
-
15.1
-
12.4
-
16.4
-20
-15
-10
-5
0
5
10
-3
-2,5
-2
-1,5
-1
-0,5
0
0,5



↓ CRP

Adiponectin